
Back Elektrochemiese sel Afrikaans خلية كهركيميائية Arabic Cel·la electroquímica Catalan Elektrochemische Zelle German Ηλεκτροχημικό στοιχείο Greek Elektrokemia ĉelo Esperanto Celda electroquímica Spanish Keemiline vooluallikas Estonian پیل برقشیمیایی Persian Cellule électrochimique French
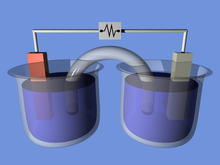
An electrochemical cell is a device that generates electrical energy from chemical reactions. Electrical energy can also be applied to these cells to cause chemical reactions to occur.[1] Electrochemical cells that generate an electric current are called voltaic or galvanic cells and those that generate chemical reactions, via electrolysis for example, are called electrolytic cells.[2]
Both galvanic and electrolytic cells can be thought of as having two half-cells: consisting of separate oxidation and reduction reactions.
When one or more electrochemical cells are connected in parallel or series they make a battery. Primary cells are single use batteries.
- ^ Wenzel, Thomas J. (July 30, 2013). "Douglas A. Skoog, Donald M. West, F. James Holler, and Stanley R. Crouch: Fundamentals of analytical chemistry, 9th ed., international ed". Analytical and Bioanalytical Chemistry. 405 (25): 412–432. doi:10.1007/s00216-013-7242-1. ISSN 1618-2642. S2CID 94566587.
- ^ Wendt, Hartmut; Kolb, Dieter M.; Engelmann, Gerald E.; Ziegler, Jörg C. (October 15, 2011), "Electrochemistry, 1. Fundamentals", in Wiley-VCH Verlag GmbH & Co. KGaA (ed.), Ullmann's Encyclopedia of Industrial Chemistry, Weinheim, Germany: Wiley-VCH Verlag GmbH & Co. KGaA, pp. a09_183.pub4, doi:10.1002/14356007.a09_183.pub4, ISBN 978-3-527-30673-2, retrieved May 5, 2023