
Back مودافينيل Arabic مودافینیل AZB Modaffinil Welsh Modafinil German Μονταφινίλη Greek Modafinilo Spanish Modafiniil Estonian مدافینیل Persian Modafiniili Finnish Modafinil French
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Provigil, Alertec, Modavigil, others |
| Other names | CRL-40476; Diphenylmethyl-sulfinylacetamide |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a602016 |
| License data | |
| Pregnancy category |
|
| Dependence liability | Relatively low[2] |
| Addiction liability | Low[2] |
| Routes of administration | By mouth[3] |
| Drug class | CNS stimulant eugeroic |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 62.3% |
| Metabolism | Liver (primarily via amide hydrolysis);[9] CYP1A2, CYP2B6, CYP2C9, CYP2C19, CYP3A4, CYP3A5 involved[11] |
| Elimination half-life | 12–15 h[9] |
| Duration of action | 11.5 h[10] |
| Excretion | Urine (80%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.168.719 |
| Chemical and physical data | |
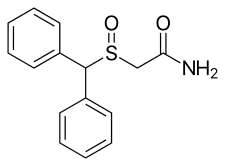
| Formula | C15H15NO2S |
| Molar mass | 273.35 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Modafinil, sold under the brand name Provigil among others, is a wakefulness-promoting medication used primarily to treat narcolepsy.[3][8][12] Modafinil is also approved for stimulating wakefulness in people with sleep apnea and shift work sleep disorder.[3] It is taken by mouth.[3][8] Modafinil is not approved by the US Food and Drug Administration (FDA) for use in people under age 17.[8]
Modafinil has potential for causing severe allergic reactions, mental (psychiatric) effects,[3] hypersensitivity, adverse interactions with prescription drugs, and misuse or abuse.[3][8][12] Modafinil may harm the fetus if taken during or two months prior to pregnancy.[13]
While modafinil is used as a cognitive enhancer or "smart drug" among healthy individuals seeking improved focus and productivity,[14][15] its use outside medical supervision raises concerns regarding potential misuse or abuse.[3][8][16] Research on the cognitive enhancement effects of modafinil in non-sleep-deprived individuals has yielded mixed results, with some studies suggesting modest improvements in attention and executive functions, while others show no significant benefits or even a decline in cognitive functions at high doses.[17][18]
- ^ a b "TGA eBS - Product and Consumer Medicine Information Licence". Archived from the original on February 28, 2024. Retrieved February 28, 2024.
- ^ a b c Cite error: The named reference
modafinil-schedule-IV-1999was invoked but never defined (see the help page). - ^ a b c d e f g "Modafinil Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. September 23, 2023. Archived from the original on March 30, 2019. Retrieved February 3, 2024.
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved October 22, 2023.
- ^ "RDC Nº 784 – Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 – Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published April 4, 2023). March 31, 2023. Archived from the original on August 3, 2023. Retrieved August 3, 2023.
- ^ "Modafinil Product information". Health Canada. April 25, 2012. Archived from the original on June 10, 2022. Retrieved June 10, 2022.
- ^ "Modafinil Provigil 100 mg Tablets – Summary of Product Characteristics (SMPC) – (Emc)". emc. June 9, 2021. Archived from the original on December 2, 2023. Retrieved December 1, 2023.
- ^ a b c d e f "Provigil- modafinil tablet". DailyMed. November 30, 2018. Archived from the original on June 10, 2022. Retrieved June 10, 2022.
- ^ a b Robertson P, Hellriegel ET (2003). "Clinical pharmacokinetic profile of modafinil". Clinical Pharmacokinetics. 42 (2): 123–137. doi:10.2165/00003088-200342020-00002. PMID 12537513. S2CID 1266677.
- ^ Cite error: The named reference
pmid22375280was invoked but never defined (see the help page). - ^ Robertson P, DeCory HH, Madan A, Parkinson A (June 2000). "In vitro inhibition and induction of human hepatic cytochrome P450 enzymes by modafinil". Drug Metabolism and Disposition. 28 (6): 664–671. PMID 10820139.
- ^ a b "Modafinil". MedlinePlus. US National Library of Medicine. February 15, 2016. Archived from the original on December 7, 2023. Retrieved February 3, 2024.
- ^ Kaplan S, Braverman DL, Frishman I, Bartov N (February 2021). "Pregnancy and Fetal Outcomes Following Exposure to Modafinil and Armodafinil During Pregnancy". JAMA Internal Medicine. 181 (2): 275–277. doi:10.1001/jamainternmed.2020.4009. PMC 7573789. PMID 33074297.
- ^ Cite error: The named reference
economist-2008-05-22was invoked but never defined (see the help page). - ^ Cite error: The named reference
Slotnik-2017was invoked but never defined (see the help page). - ^ Cite error: The named reference
pmid30285371was invoked but never defined (see the help page). - ^ Cite error: The named reference
pmid36056861was invoked but never defined (see the help page). - ^ Cite error: The named reference
pmid32341841was invoked but never defined (see the help page).