
Back سيليجيلين Arabic سلژیلین AZB Selegilin Welsh Selegilin German Selegilina Spanish سلژیلین Persian Selegiliini Finnish Sélégiline French סלג'ילין HE Szelegilin Hungarian
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /səˈlɛdʒɪliːn/ sə-LEJ-i-leen |
| Trade names | Eldepryl, Jumex, Zelapar, Emsam, others[1] |
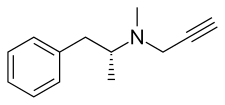
| Other names | L-Deprenyl; (R)-(–)-N,α-Dimethyl-N-2-propynylphenethylamine; (R)-(–)-N-Methyl-N-2-propynylamphetamine; (R)-(–)-N-2-propynylmethamphetamine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a697046 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, transdermal (patch) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 10% (oral), 73% (patch) |
| Protein binding | 94% |
| Metabolism | Intestines and liver |
| Metabolites | N-Desmethylselegiline, levoamphetamine, levomethamphetamine |
| Elimination half-life | 1.5–3.5 hours (oral),[3] 18–25 hours (transdermal)[4] |
| Excretion | Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.109.269 |
| Chemical and physical data | |
| Formula | C13H17N |
| Molar mass | 187.286 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Selegiline, also known as L-deprenyl and sold under the brand names Eldepryl, Emsam, Selgin, among other names, is a medication which is used in the treatment of Parkinson's disease and major depressive disorder.[1] It is provided in the form of a capsule or tablet taken by mouth or orally disintegrating tablets taken on the tongue for Parkinson's disease and as a patch applied to skin for depression.
Selegiline acts as a monoamine oxidase inhibitor, and increases levels of monoamine neurotransmitters in the brain. At typical clinical doses used for Parkinson's disease, selegiline is a selective and irreversible inhibitor of monoamine oxidase B (MAO-B), increasing levels of dopamine in the brain. In larger doses (more than 20 mg/day), it loses its specificity for MAO-B and also inhibits MAO-A, which increases serotonin and norepinephrine levels in the brain.
- ^ a b "Selegiline". Drugs.com. Retrieved February 7, 2016.
- ^ Anvisa (March 31, 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published April 4, 2023). Archived from the original on August 3, 2023. Retrieved August 16, 2023.
- ^ "Résumé des Caractéristiques du Produit".
- ^ https://www.accessdata.fda.gov/drugsatfda_docs/label/2007/021336s002lbl.pdf [bare URL PDF]