Back جالكتوز Arabic Qalaktoza Azerbaijani قالاکتوز AZB Галактоза Byelorussian Галяктоза BE-X-OLD Галактоза Bulgarian Galaktoza BS Galactosa Catalan گالاکتۆز CKB Galaktóza Czech
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
Galactose
galacto-Hexose[1] | |||
| Systematic IUPAC name
(2R,3S,4S,5R,6)-2,3,4,5,6-Pentahydroxyhexanal | |||
| Other names
Brain sugar
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 1724619 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| KEGG | |||
| MeSH | Galactose | ||
PubChem CID
|
|||
| UNII | |||
| |||
| |||
| Properties | |||
| C6H12O6 | |||
| Molar mass | 180.156 g·mol−1 | ||
| Appearance | White solid[2] | ||
| Odor | Odorless[2] | ||
| Density | 1.5 g/cm3[2] | ||
| Melting point | 168–170 °C (334–338 °F; 441–443 K)[2] | ||
| 650 g/L (20 °C)[2] | |||
| −103.00·10−6 cm3/mol | |||
| Pharmacology | |||
| V04CE01 (WHO) V08DA02 (WHO) (microparticles) | |||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
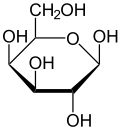
Galactose (/ɡəˈlæktoʊs/, galacto- + -ose, 'milk sugar'), sometimes abbreviated Gal, is a monosaccharide sugar that is about as sweet as glucose, and about 65% as sweet as sucrose.[3] It is an aldohexose and a C-4 epimer of glucose.[4] A galactose molecule linked with a glucose molecule forms a lactose molecule.
Galactan is a polymeric form of galactose found in hemicellulose, and forming the core of the galactans, a class of natural polymeric carbohydrates.[5]
D-Galactose is also known as brain sugar since it is a component of glycoproteins (oligosaccharide-protein compounds) found in nerve tissue.[6]
- ^ "Appendix".
- ^ a b c d e Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ^ Spillane WJ (2006-07-17). Optimising Sweet Taste in Foods. Woodhead Publishing. p. 264. ISBN 9781845691646.
- ^ Kalsi PS (2007). Organic Reactions Stereochemistry And Mechanism (Through Solved Problems). New Age International. p. 43. ISBN 9788122417661.
- ^ Zanetti M, Capra DJ (2003-09-02). The Antibodies. CRC Press. p. 78. ISBN 9780203216514.
- ^ "16.3 Important Hexoses | The Basics of General, Organic, and Biological Chemistry". courses.lumenlearning.com. Retrieved 2022-05-06.