
Back سوماتريبتان Arabic سوماتریپتان AZB Sumatriptan Catalan Sumatriptan German Σουματριπτάνη Greek Sumatriptán Spanish سوماتریپتان Persian Sumatriptaani Finnish Sumatriptan French סומטריפטן HE
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Imitrex, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601116 |
| License data |
|
| Routes of administration | By mouth, subcutaneous, intranasal, transdermal |
| Drug class | Antimigraine agent; Triptan; Serotonin 5-HT1B, 5-HT1D receptor, and 5-HT1F receptor agonist |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 15% (oral); 96% (subcutaneous) |
| Protein binding | 14–21% |
| Metabolism | Monoamine oxidase (MAO) |
| Elimination half-life | 2.5 hours |
| Excretion | 60% urine; 40% feces |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.130.518 |
| Chemical and physical data | |
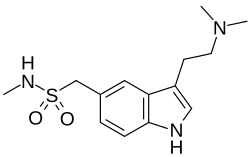
| Formula | C14H21N3O2S |
| Molar mass | 295.40 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Sumatriptan, sold under the brand name Imitrex among others, is a medication used to treat migraine headaches and cluster headaches.[4][1] It is taken orally, intranasally, or by subcutaneous injection.[5] Therapeutic effects generally occur within three hours.[5] Sumatriptan is a serotonin (5-HT1B/1D) receptor agonist (triptan).[1]
The drug acts as a serotonin 5-HT1B, 5-HT1D, and 5-HT1F receptor agonism[6][7] and its common side effects include chest pressure, fatigue, vomiting, tingling, and vertigo. Serious side effects may include serotonin syndrome, heart attack, stroke, and seizures. With excessive use, medication overuse headaches may occur.[5] It is unclear if use during pregnancy or breastfeeding is safe.[8] The mechanism of action is not entirely clear. It is in the triptan class of medications.[5]
Sumatriptan was patented in 1982 and approved for medical use in 1992.[1][9] It is on the World Health Organization's List of Essential Medicines.[10] It is available as a generic medication.[4] In 2022, it was the 95th most commonly prescribed medication in the United States, with more than 6 million prescriptions.[11][12] It is also available as the combination product sumatriptan/naproxen.[13]
- ^ a b c d "Imitrex- sumatriptan injection". DailyMed. 8 February 2023. Retrieved 6 July 2025.
- ^ "Imitrex- sumatriptan spray". DailyMed. 22 March 2024. Retrieved 6 July 2025.
- ^ "Imitrex- sumatriptan tablet, film coated". DailyMed. 21 February 2024. Retrieved 6 July 2025.
- ^ a b British National Formulary: BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 474. ISBN 9780857113382.
- ^ a b c d "Sumatriptan Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 3 March 2019.
- ^ Cite error: The named reference
Rubio-BeltránLabastida-RamírezVillalón2018was invoked but never defined (see the help page). - ^ Syed YY (January 2016). "Sumatriptan/Naproxen Sodium: A Review in Migraine". Drugs. 76 (1): 111–121. doi:10.1007/s40265-015-0521-8. PMID 26628293. S2CID 25060147.
- ^ "Sumatriptan Use During Pregnancy". Drugs.com. Retrieved 3 March 2019.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 531. ISBN 9783527607495.
- ^ World Health Organization (2021). World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ "The Top 300 of 2022". ClinCalc. Archived from the original on 30 August 2024. Retrieved 30 August 2024.
- ^ "Sumatriptan Drug Usage Statistics, United States, 2013 - 2022". ClinCalc. Retrieved 30 August 2024.
- ^ "Treximet- sumatriptan succinate and naproxen sodium tablet, film coated". DailyMed. 23 December 2024. Retrieved 6 July 2025.