Medicines and Healthcare products Regulatory Agency - Activity on GOV.UK
“Electrical Safety – An Overview” (Tamil) Electricity is undoubtedly one of the most basic needs in modern life, but electrical hazards are very dangerous, sometimes we ...
Show Comments
Electrical Safety | KS2 Science | STEM and Beyond Do you like learning about electricity? Let's look at electrical safety! In this video, we will find out more about the dangers of ...
Medicines and Healthcare products Regulatory Agency - Activity on GOV.UK
Medicines and Healthcare products Regulatory Agency - Activity on GOV.UK
Public Health England - Activity on GOV.UK
Electricity! ⚡ Electrical Safety with The Wiggles and Queensland Electrical Safety Office The Wiggles have partnered with the Queensland Government's Electrical Safety Office to deliver life-saving electrical safety ...
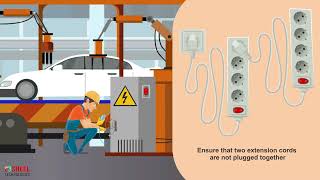
10 Electrical Safety Rules Training Video in English || Hazards & Precautions #electricalsafety Electrically powered equipment can pose a significant hazard to workers, particularly when mishandled or not maintained.
Preventing Workplace Hazards: Basic Electrical Safety Training Electricity powers our world, but without proper precautions, it can be a silent and deadly threat. Each year, thousands of ...
Public Health England - Activity on GOV.UK
Public Health England - Activity on GOV.UK
OSHA Electrical Safety: How To Prevent Electrical Hazards. Automate Safety Talks and Save Time - https://safelyio.com/ Generate Free safety toolbox talks PDFs ...
Medicines and Healthcare products Regulatory Agency - Activity on GOV.UK
Medicines and Healthcare products Regulatory Agency - Activity on GOV.UK
Medicines and Healthcare products Regulatory Agency - Activity on GOV.UK
Medicines and Healthcare products Regulatory Agency - Activity on GOV.UK
Medicines and Healthcare products Regulatory Agency - Activity on GOV.UK
Medicines and Healthcare products Regulatory Agency - Activity on GOV.UK
Load More